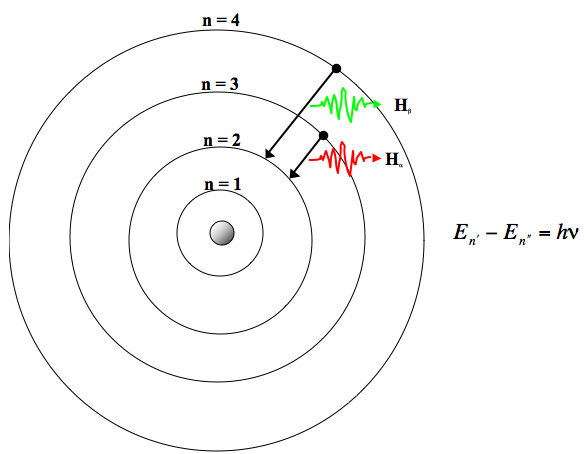
Es beschreibt, dass die Elektronen den Atomkern auf sogenannten Kreisbahnen umkreisen. Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron. Das Bohrsche Atommodell ist ein Modell, das erklärt, wie ein Atom aufgebaut ist. The numbers of particles in an atom can be calculated from its atomic number and mass. 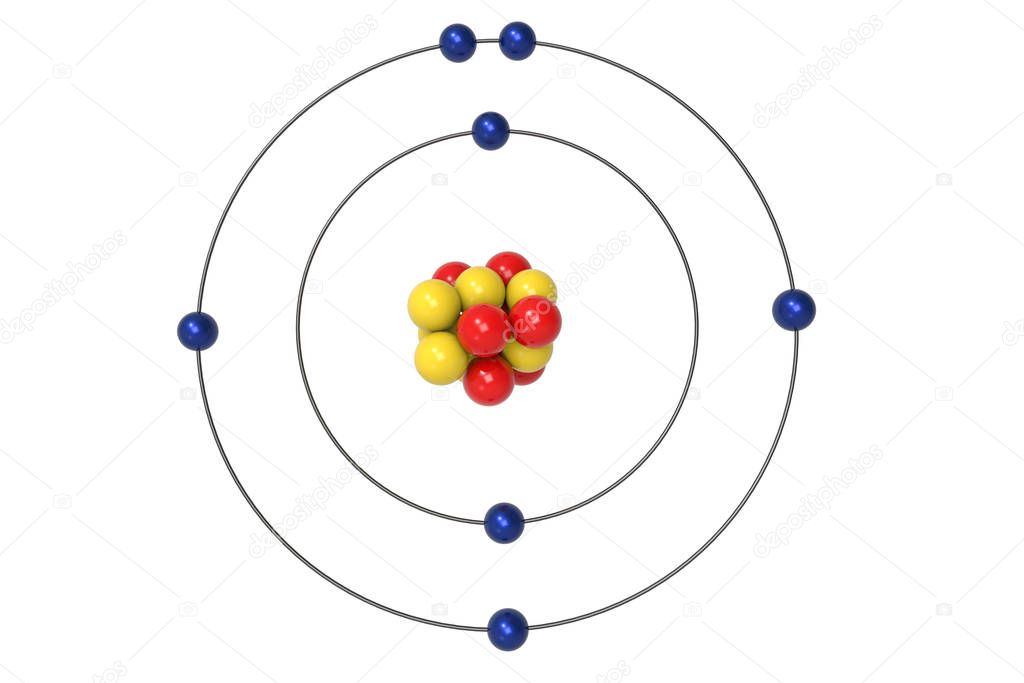
Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. Gambar model atom Niels Bohr (Arsip Zenius) Kulit paling dalam atau yang paling dekat dengan inti atom memiliki tingkat energi paling rendah, sedangkan yang memiliki tingkat energi paling tinggi terletak pada kulit paling luar atau paling jauh dari inti atom. The Bohr Model is a modification of the Rutherford Model that explains the Rydberg formula for the emission lines of atomic hydrogen. The atomic model consists of a nucleus containing protons and neutrons, surrounded by electrons in shells. 1 El model de Bohr considera que làtom està format per un nucli atòmic molt petit que conté tota la càrrega positiva i gairebé tota la massa de làtom. In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Since Bohr’s model involved only a single electron, it could also be applied to the single electron ions He +, Li 2 +, Be 3 +, and so forth, which differ from hydrogen only in their nuclear charges, and so one-electron atoms and ions are collectively referred to as hydrogen-like atoms. El model atòmic de Bohr és una teoria sobre la constitució dels àtoms, cal citació proposada pel físic danès Niels Bohr lany 1913. The Bohr model shows the atom as a small, positively charged nucleus surrounded by orbiting electrons. Where \(n_1\) and \(n_2\) are positive integers, \(n_2 > n_1\), and \( \Re \) the Rydberg constant, has a value of 1.09737 × 10 7 m −1. Bohrs greatest contribution to modern physics was the atomic model.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
